Old methods prove true for studying proteins
12 October, 2020
A fresh new look at an old technique in protein biochemistry has shown that it should be reintroduced to the spectroscopy toolkit.
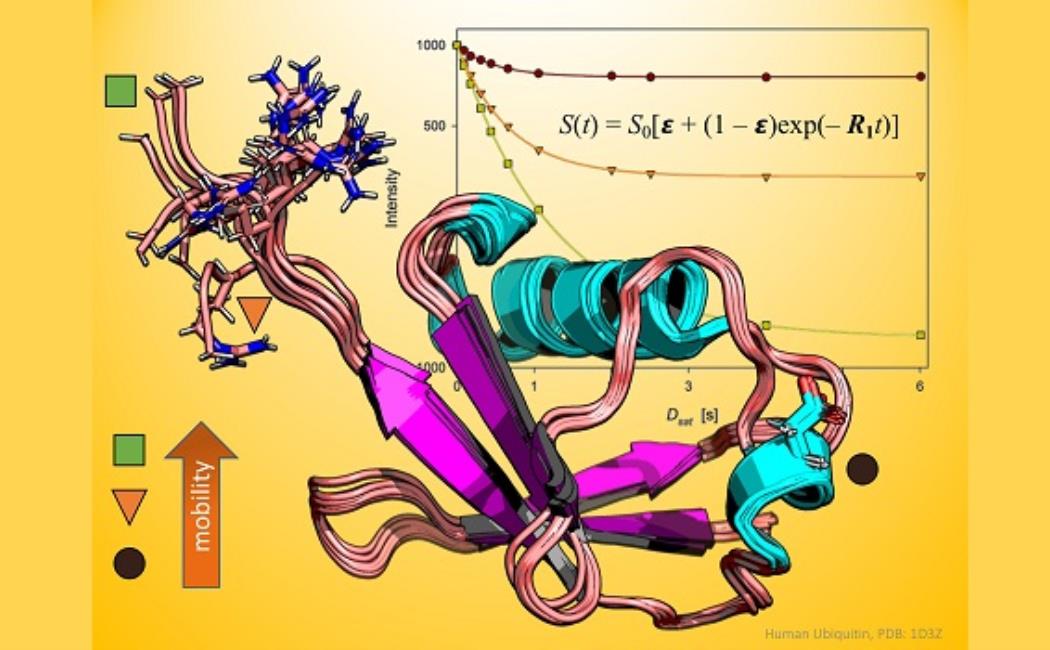
For decades, scientists have used nuclear magnetic resonance (NMR) spectroscopy to probe the molecular motions of proteins on various timescales. This technique has revealed aspects of enzyme reactions, protein folding and other biological processes, all on an atomic scale.
Typically, spectroscopists will gauge the rotation of NMR-active atoms in the protein backbone with and without proton irradiation to calculate a ratio known as a steady-state nuclear Overhauser effect (NOE); however, it was not always done this way.
Click here to read the full story
Image: Dynamic nuclear Overhauser effect accurately tracks the biological motions of proteins.
© 2020 Łukasz Jaremko and Vladlena Kharchenko